Dental units serve as main workstations in dental practices. Many patients receive care there each day. Good dental unit disinfection forms the base of infection control steps. These steps protect patients and staff from cross-contamination. Dental chair units include parts such as waterlines, suction systems, instrument trays, and operator surfaces. These parts often touch oral fluids. Without regular disinfection, these areas can build up biofilms and pathogens. Such buildup lowers clinical safety standards.
 The Basics of Dental Infection Control
The Basics of Dental Infection Control
Infection control in dental settings uses clear procedures for cleaning, disinfecting, and sterilizing surfaces and instruments. Dental professionals follow protocols that reduce microbes on high-touch zones of dental chair units. These protocols depend on EPA-registered disinfectants, steady personal protective equipment use, and close following of manufacturer instructions. Staff training helps make sure everyone carries out disinfection steps the same way during procedures.
Dental unit waterlines create a special problem. Stagnant water helps bacteria grow. Regular flushing plus chemical treatments keeps water quality at levels that meet microbiological standards for dental procedures. Suction lines need the same care to stop contaminant aerosolization.
Poor disinfection of dental chair units leads to serious problems. Bloodborne pathogens, such as hepatitis B, hepatitis C, and HIV, can spread through dirty instruments or surfaces. Respiratory infections move more easily in places with weak hygiene controls. Long contact with dirty waterlines links to higher rates of opportunistic infections in patients who are at risk.
Regulatory authorities set strict rules for dental facilities. Violations lead to shutdowns, fines, and legal risks. Solid disinfection protocols reduce these dangers. They also allow steady delivery of high-quality care.
Modern disinfection dental chair units include built-in systems. These systems automate and improve hygiene processes. The units have features that handle waterline contamination, surface treatment, and instrument compatibility. Proper maintenance routines extend equipment life. They also keep the performance levels needed in professional clinics.
Suitable dental unit cleaners target biofilm in tubing and reservoirs. High-quality formulas clear organic deposits. They do this without damage to sensitive parts. Clinics use layered schedules. These cover quick post-patient surface treatment, daily waterline flushing, weekly deep cleaning, and regular professional servicing.
A standard maintenance framework includes these steps:
Steady use stops contaminant buildup. It also maintains dental chair unit integrity.
Dental practices review disinfectant categories. They choose ones that fit operational needs. Chemical agents give broad action against bacteria, viruses, and fungi. Natural alternatives appeal to facilities that want less chemical exposure. These work if they pass efficacy tests and show similar performance.
Chemical disinfectants that use alcohols, quaternary ammonium compounds, or chlorine derivatives offer fast pathogen reduction on dental chair unit surfaces. These fit well with automated systems in newer models. Proper dilution and contact times improve results. They also limit material wear.
Natural disinfectants come from plant extracts or enzymes. They have lower toxicity. Their use needs proof of efficacy. This proof must confirm pathogen reduction equal to standard options. Hybrid approaches sometimes combine both types. This creates balance in safety and effectiveness for all unit components.
Delivery methods affect disinfection consistency and efficiency. Automated systems in disinfection dental chair units provide programmed dosing and flushing cycles. These reduce manual mistakes. Manual methods still help with detailed cleaning of complex surfaces during maintenance.
Integrated platforms often mix automated waterline treatment with manual surface protocols. This method simplifies workflows. It also ensures full coverage of critical zones.
 Dental sterilization kills all microbial forms on instruments that touch tissue or blood. Methods like autoclaving, chemical vapor, and dry heat fit different instrument materials. Surface disinfection adds to sterilization. It manages non-critical dental chair unit elements.
Dental sterilization kills all microbial forms on instruments that touch tissue or blood. Methods like autoclaving, chemical vapor, and dry heat fit different instrument materials. Surface disinfection adds to sterilization. It manages non-critical dental chair unit elements.
Core steps in the sterilization cycle include pre-cleaning to remove debris, inspection and packaging, validated sterilization application, controlled storage, and routine surface disinfection between patients. These steps together cut cross-contamination risks. They also build patient confidence in safety.
Organized protocols create repeatable hygiene standards in dental operations. Facilities build schedules based on patient volume and equipment specifications. Staff training strengthens technique and clinical reasons for each action.
Schedules group tasks by frequency. This keeps hygiene uniform. Post-patient routines focus on quick surface treatment. Daily activities handle water and suction systems. Weekly tasks cover detailed handpiece and accessory reviews. Monthly checks track supplies and compliance.
Training programs mix teaching with hands-on practice. Regular refreshers update teams on guidelines and unit-specific features. Competency evaluations spot areas for improvement. Accountability encourages early detection of hygiene gaps.
Facilities check disinfection systems on several performance aspects. Efficacy data reveals pathogen reduction rates. Safety profiles confirm exposure limits and equipment durability. Interface design influences team adoption. Compatibility with existing setups prevents costly changes.
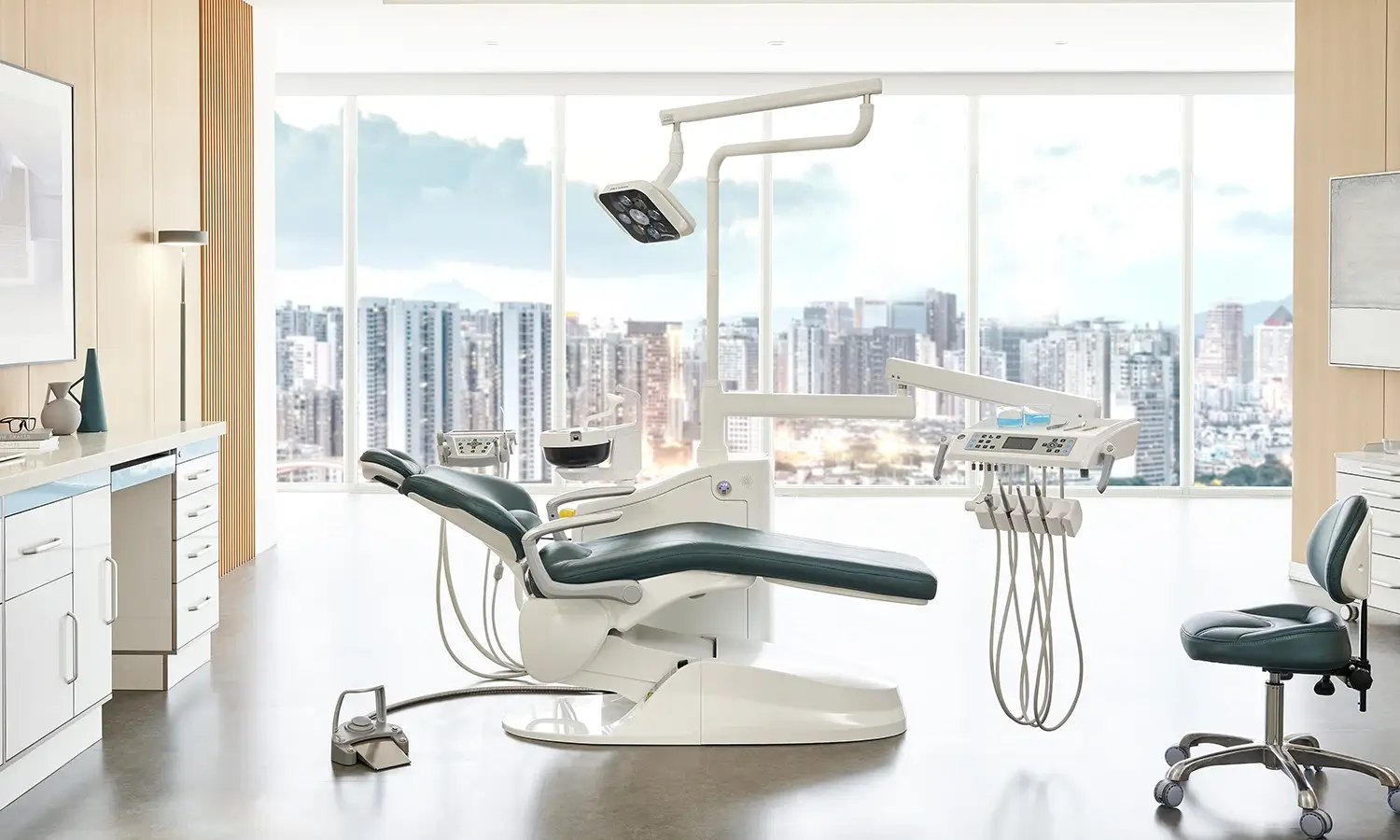
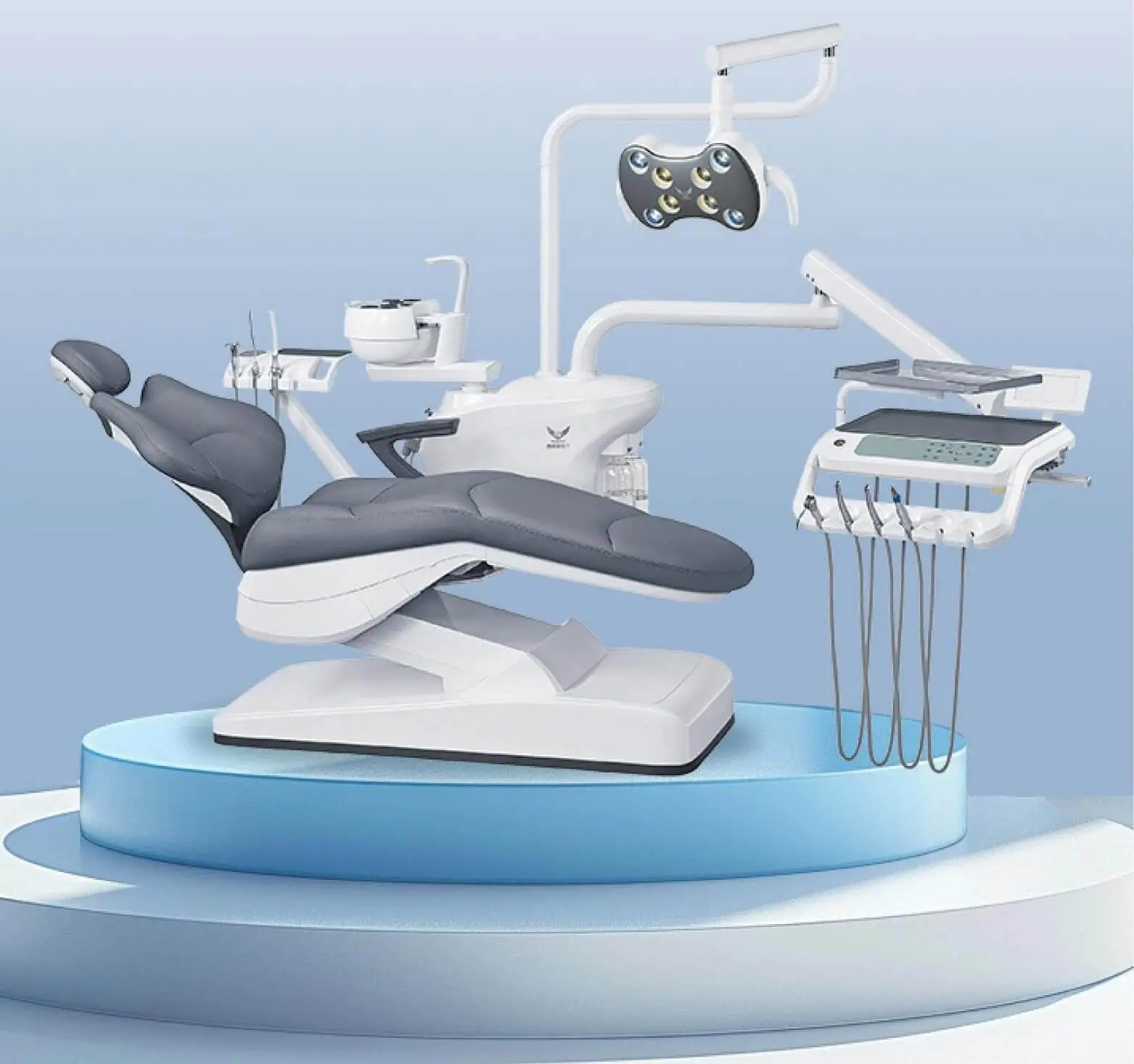
Modern disinfection dental chair units add smart technologies. These improve infection control. The GD-S800 model shows these advances. It offers one-click full pipeline disinfection. This treats waterlines, handpiece tubing, three-way syringe lines, and high/low suction circuits at the same time. Automatic pulse line sterilization sends disinfectant in controlled pulses. The pulses reach difficult internal areas.
A dual-bottle water storage system permits easy switching. It moves between distilled water for daily use and disinfectant solutions for hygiene cycles. This setup allows complete disinfection of the entire waterline and suction circuits. It removes up to 99% of harmful bacteria. Imported water and air tubes plus a high-quality Taiwan motor ensure reliable performance under repeated chemical exposure.
Additional design elements include nine memory positions for patient-specific adjustments, a butterfly-shaped metal backrest for ergonomic support, and one-click controls for water, air, and power supplies. These features lower operational complexity. They still keep strict hygiene standards.
Waterlines in dental chair units require focused management. This blocks biofilm development. Advanced models carry out automated shock treatments and continuous low-level disinfection. Built-in monitoring aids timely maintenance. Daily flushing together with periodic chemical interventions keeps water quality inside industry benchmarks. Replaceable filters catch particulates. Professional servicing maintains pump, valve, and sensor functionality.
Disinfection covers all external surfaces of the dental chair unit. Materials stand up to repeated hospital-grade disinfectant application. They do not degrade. Seamless upholstery and smooth contours make cleaning easier. They prevent liquid buildup. Instrument delivery systems support automated disinfection cycles. These preserve sterility between uses.
Quality disinfection dental chair units bring returns. They lead to fewer infection events and better efficiency. Fewer interruptions allow higher patient throughput. Strong safety records boost retention and referrals. Regulatory adherence cuts penalty risks. Total ownership costs cover acquisition, training, maintenance, and savings from avoided incidents. Strong construction extends service intervals. It also lowers repair frequency.
Practices meet obstacles such as time limits in busy settings, equipment complexity that needs special methods, and staff changes from turnover. Automated features and simple controls make processes easier. Standardized procedures reduce differences in execution. Documentation tools track compliance. They also point out trends that need attention.
Dental clinics and distributors looking for reliable disinfection dental chair units gain advantages through collaboration with an established manufacturer and supplier. Gladent, a professional factory specializing in dental equipment since 2013, produces the GD-S800 Disinfection Dental Unit with one-click full pipeline disinfection, automatic pulse sterilization, dual-bottle systems, and compatibility with disinfectants that remove 99% of pipeline bacteria. The model includes imported components, ergonomic design, nine memory positions, and CE certification.
As a dedicated manufacturer and supplier, Gladent provides competitive wholesale pricing, customization, and support for bulk orders or dealerships. Dental equipment providers contact the factory directly for specifications, quotations, and partnership details. Reach out via email at gladent08@dental-chair.cn or telephone +86-757-81267151 to secure supply from the production source.
Clinics review automated disinfection functions, waterline management, material compatibility, maintenance ease, and certifications such as CE.
Consistent disinfection prevents cross-contamination, protects health, ensures regulatory compliance, and upholds professional standards.
Surfaces receive treatment after each patient. Waterlines and suction lines undergo daily flushing. Deeper disinfection follows manufacturer guidelines, typically weekly.
Transmission of pathogens, higher infection rates, equipment damage, regulatory violations, and reputational harm.
Natural options serve as alternatives when efficacy against dental pathogens is validated. Many practices combine them with chemical agents for specific applications.